
1. Generic Name
Lobeglitazone Sulfate Tablets 0.5 mg
2. Qualitative and Quantitative Composition
Each Uncoated Tablet Contains:
Lobeglitazone Sulfate ……………………………... 0.5 mg
3. Dosage Form and Strength
Uncoated Tablet; 0.5 mg


Lobeglitazone Sulfate Tablets 0.5 mg
Each Uncoated Tablet Contains:
Lobeglitazone Sulfate ……………………………... 0.5 mg
Uncoated Tablet; 0.5 mg
Indicated for treatment of adult type 2 diabetes mellitus patients:
As monotherapy
Posology
The recommended oral dosage is 1 tablet once daily.
Patients with hepatic impairment
At the beginning of treatment, if the patient shows clinical evidence of active liver disease or increased serum transaminase levels (more than 2.5 times the upper limit of ALT or AST), therapy with this drug should not be initiated.
Note
Liver enzyme monitoring is recommended in all patients before and on a regular basis after starting Lobeglitazone Tablet 0.5 mg therapy.
Patients with renal impairment
Dosage adjustment is not necessary for patients with mild to moderate renal impairment.
Method of administration: For oral use only.
Thiazolidinedione drugs, including Lobeglitazone, may cause or worsen congestive heart failure in some patients and should be administered with the care of a doctor.
After starting treatment with this drug, the patient should be carefully monitored for symptoms and signs of heart failure (including excessive and rapid weight change, dyspnea, and swelling). If these symptoms and signs appear, tests (e.g., echocardiography, chest x-ray, electrocardiogram, related blood tests (NT-proBNP), etc.) are performed to evaluate them. Heart failure should be managed according to current standard treatment regimens, and discontinuation of this drug should be considered.
Patients with severe heart failure (patients with New York Heart Association (NYHA) Class 1 to 4 heart conditions) should not start treatment with this drug. Lobeglitazone Tablet 0.5 mg is not recommended for patients with symptomatic heart failure.
Heart failure and other actions on the heart: Lobeglitazone, like other thiazolidinediones, can cause fluid retention when administered alone or in combination with other antidiabetic drug such as insulin. Fluid retention can cause or worsen heart failure. Patients should be observed for signs or symptoms of heart failure.
Administer carefully to the following patients
This drug did not inhibit the p450 enzyme at clinically relevant doses. It is metabolized by CYP 2C19, 2D6, and 3A4.
Metformin: When 0.5 mg of this drug was administered with metformin 1000 mg, no clinically significant interaction.
Glimepiride: No clinically significant effect on pharmacokinetics of glimepiride.
Amlodipine: Did not significantly affect each other's pharmacokinetic properties.
Ketoconazole: Increased exposure level of this drug by about 33%.
Warfarin: Did not affect each other's pharmacokinetic properties.
Sitagliptin, Empagliflozin, Dapagliflozin: No significant pharmacokinetic interactions.
Pregnancy: There is no appropriate clinical test result for pregnant women, so use in pregnant women is not recommended.
Lactation: It is not known whether this drug is secreted in human breast milk. Lobeglitazone has been reported to be secreted into milk in lactating rats, so it should not be administered to nursing mothers.
No studies on the effects on the ability to drive and use machines have been performed. However, patients who experience visual disturbance should be cautious when driving or using machines.
Table 1. Adverse reactions reported in more than 1% of patients in the placebo-controlled monotherapy clinical trials and active drug-controlled metformin combination therapy clinical trials in the group treated with 0.5mg of Lobeglitazone tablet (regardless of the researcher's causal assessment)
| Adverse reaction | Placebo N=58 (%) | Lobeglitazone tablet 0.5 mg N=112 (%) | Pioglitazone 15 mg + Metformin N=125 (%) | Lobeglitazone tablet 0.5 mg + Metformin N=128 (%) |
|---|---|---|---|---|
| Edema | 2 (3.45) | 7 (6.25) | 2 (1.60) | 5 (3.91) |
| Headache | 2 (3.45) | 3 (2.68) | 0 (0.00) | 0 (0.00) |
| Chest pain | 1 (1.72) | 2 (1.79) | 0 (0.00) | 0 (0.00) |
| Tooth decay | 1 (1.72) | 2 (1.79) | 0 (0.00) | 0 (0.00) |
| Diarrhea | 0 (0.00) | 1 (0.89) | 1 (0.80) | 3 (2.34) |
| Cold | 0 (0.00) | 6 (5.36) | 10 (8.00) | 6 (4.69) |
| Upper respiratory infection | 3 (5.17) | 2 (1.79) | 2 (1.60) | 2 (1.56) |
| Hyperglycemia | 4 (6.90) | 3 (2.68) | 2 (1.60) | 1 (0.78) |
| Itching | 1 (1.72) | 2 (1.79) | 0 (0.00) | 2 (1.56) |
| Fracture | 0 (0.00) | 0 (0.00) | 3 (2.40) | 3 (2.34) |
| ALT increase | 0 (0.00) | 2 (1.79) | 0 (0.00) | 0 (0.00) |
| AST increase | 0 (0.00) | 2 (1.79) | 0 (0.00) | 0 (0.00) |
| Dizziness | 0 (0.00) | 3 (2.68) | 0 (0.00) | 3 (2.34) |
| Anemia | 0 (0.00) | 3 (2.68) | 0 (0.00) | 2 (1.56) |
Monotherapy for 52-week extended trial: Among the adverse reactions reported: reflux esophagitis (2 patients, 3.13%), upper respiratory infections (5 persons, 7.81%), dizziness (2 persons, 3.13%).
Edema: In the 24-week monotherapy clinical trial, incidence of edema was 3.45% (2 patients) in placebo and 6.25% (7 patients) in lobeglitazone 0.5mg group.
Weight gain: In 24-week monotherapy clinical trial, placebo group decreased by about 0.63 kg, Lobeglitazone 0.5 mg-administered group increased by about 0.89 kg.
Hypoglycemia: Lobeglitazone tablet did not show hypoglycemia in monotherapy trial. In combination therapy, reported in 1 patient (0.78%) in Lobeglitazone 0.5mg group.
Reporting of Suspected Adverse Reactions: Healthcare professionals are asked to report any suspected adverse reactions via email to: medico@zorvia.com
Data on overdose in humans are limited. In clinical trials, the drug was administered orally up to 7 mg for 4 days and was well tolerated. In case of overdose, appropriate adjuvant treatment is given according to the patient's clinical condition. The drug has a high rate of protein binding, so it is not eliminated by hemodialysis.
Lobeglitazone is a thiazolidinedione class agent that exerts its pharmacological effect through activation of the peroxisome proliferator–activated receptor gamma (PPAR γ). It improves insulin sensitivity by enhancing insulin dependent glucose uptake and utilization in peripheral tissues, particularly skeletal muscle, without stimulating insulin secretion from pancreatic β cells.
Single dose and multiple dose pharmacokinetic studies in healthy volunteer demonstrated that lobeglitazone is rapidly and nearly completely absorbed after oral administration reaching peak plasma levels (Tmax) at 1.0 to 3.0 hours. It is eliminated mostly by metabolism with negligible urine excretion and has a half-life of 7.8 to 9.8 hours. There was dose proportional increase in plasma concentrations for doses 0.5 mg up to 2 mg. Lobeglitazone is highly bound to plasma proteins in-vitro by >99%, mostly albumin. This drug is excreted mainly from the kidney as an unchanged drug.
(1) Carcinogenicity: As a result of the carcinogenicity test for 2 years, there was no carcinogenicity in mice when administered orally up to 6 mg/kg/day.
(2) Genotoxicity: Genotoxicity studies did not show mutagenicity.
(3) Repeated administration toxicity: Diffuse hypertrophy of the myocardium was observed in rats.
(4) Reproductive development toxicity: There was no teratogenicity when the drug was administered up to 0.8 mg/kg in rats and 90 mg/kg in rabbits.
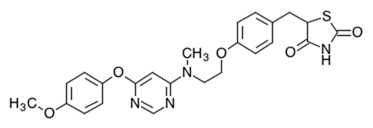
The chemical name for Lobeglitazone Sulphate is 5-[[4-[2-[[6-(4-methoxyphenoxy) pyrimidin-4-yl]-methylamino] ethoxy] phenyl] methyl]-1, 3-thiazolidine-2, 4-dione; sulfuric acid.
Molecular formula of Lobeglitazone Sulphate is C₂₄H₂₄N₄O₆S·H₂SO₄ and a molecular weight of 578.6 g/mol.
Not applicable.
Refer on the pack.
Refer on the pack.
Store protected from light & moisture at a temperature not exceeding 30°C. Keep out of reach of children.
24th March 2026