1.0 Generic Name
Empagliflozin Tablets 10mg/ 25 mg
2.0 Qualitative and Quantitative Composition
Each film coated tablets contains:
Empagliflozin………….10mg/ 25mg
Excipients…………………q.s.
3.0 Dosage Form and Strength
Tablet 10 mg / 25 mg


Empagliflozin Tablets 10mg/ 25 mg
Each film coated tablets contains:
Empagliflozin………….10mg/ 25mg
Excipients…………………q.s.
Tablet 10 mg / 25 mg
Table 1: Presents the recommended dosage of EMPLIC in adult
|
Indication |
Recommended Dosage |
|
· As an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes Mellitus. |
· 10 mg orally once daily in the morning, taken with or without food. · For additional glycemic control, it may increase to 25 mg orally once daily in patients tolerating 10 mg once daily. |
|
· To reduce the risk of cardiovascular death plus hospitalization for heart failure in adults with heart failure and reduced independent of left ventricular ejection fraction. The benefits are clearly evident in adults with reduced, mid-range, or preserved ejection fraction. · To reduce the risk of sustained decline in EGFR, end-stage kidney disease, renal death, cardiovascular death, and all cause hospitalization in adults with chronic kidney disease · Indicated in adult patients with heart failure (Nyha class ii-iv) and reduced ejection fraction with or without type 2 diabetes mellitus: to reduce the risk of cardiovascular death and hospitalization for heart failure to slow kidney function decline. |
· 10 mg orally once daily in the morning, taken with or without food. |
Hypersensitivity to the empagliflozin or to any of the excipients
Diabetic Ketoacidosis: Empagliflozin increases the risk of diabetic ketoacidosis, a life-threatening event, including fatal ketoacidosis. Empagliflozin is not indicated for glycemic control in patients with type 1 diabetes mellitus. Risk factors include type 2 diabetes mellitus, pancreatic disorders, under-insulinization, acute illness, reduced caloric intake, surgery, volume depletion, and alcohol abuse. Signs and symptoms include nausea, vomiting, abdominal pain, malaise, and shortness of breath; blood glucose may be below those typically expected. Assess for ketoacidosis regardless of blood glucose. If suspected, discontinue Empagliflozin and treat promptly. Withhold in situations predisposing to ketoacidosis and resume when clinically stable. Educate patients on signs and symptoms.
Volume Depletion: Empagliflozin may cause intravascular volume depletion, symptomatic hypotension, or acute transient changes in creatinine. Acute kidney injury has been reported. Assess and correct volume status before initiation and monitor renal function.
Genitourinary Infections: Empagliflozin increases the risk of urinary tract infections and genital mycotic infections. Serious infections, including urosepsis, pyelonephritis, and necrotizing fasciitis of the perineum (Fournier’s gangrene), have occurred. Monitor and treat promptly; discontinue if severe infection is suspected.
Hypoglycaemia: Risk may be increased with insulin or insulin secretagogues. Consider dose reduction and educate patients on signs and symptoms.
Lower Limb Amputation: Lower limb amputations have been observed. Risk is highest in patients with a history of diabetic foot, peripheral artery disease, or diabetes. Monitor for signs and symptoms and counsel on routine preventative foot care.
Hypersensitivity Reactions: Serious hypersensitivity reactions, including angioedema, have been reported. Discontinue if occurs. Contraindicated in patients with hypersensitivity to empagliflozin or excipients.
Clinically Relevant Interactions with Empagliflozin
|
Diuretics |
|
|
Clinical Impact |
Coadministration of empagliflozin with diuretics resulted in increased urine volume and frequency of voids, which might enhance the potential for volume depletion. |
|
Intervention |
Before initiating Empagliflozin, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating Empagliflozin. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy. |
|
Insulin or Insulin Secretagogues |
|
|
Clinical Impact |
The risk of hypoglycemia is increased when Empagliflozin is used in combination with insulin secretagogues (e.g., sulfonylurea) or insulin. |
|
Intervention |
Coadministration of Empagliflozin with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower dosages of the insulin secretagogue or insulin to reduce the risk of hypoglycemia. |
|
Lithium |
|
|
Clinical Impact |
Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. |
|
Intervention |
Monitor serum lithium concentration more frequently during Emplic initiation and dosage changes. |
|
Positive Urine Glucose Test |
|
|
Clinical Impact |
SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. |
|
Intervention |
Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. |
|
Interference with 1,5-anhydroglucitol (1,5-AG) Assay |
|
|
Clinical Impact |
Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. |
|
Intervention |
Monitoring glycemic control with 1,5-AG assay is not recommended. Use alternative methods to monitor glycemic control |
Pregnancy
Risk Summary: Based on animal data showing adverse renal effects, Empagliflozin is not recommended during the second and third trimesters of pregnancy. Available data in pregnant women are not sufficient to determine a drug-associated risk for major birth defects and miscarriage. Poorly controlled diabetes in pregnancy increases maternal and fetal risks. In animals, renal pelvic and tubule dilatations were observed at exposures approximately 13-times the maximum clinical dose and were reversible.
Clinical Considerations: Poorly controlled diabetes increases the risk of diabetic ketoacidosis, pre-eclampsia, spontaneous abortions, preterm delivery, delivery complications, major birth defects, stillbirth, and macrosomia.
Data: In juvenile rats, empagliflozin caused increased kidney weights and renal tubular and pelvic dilatation at approximately 13-times the maximum clinical dose, which were not observed after recovery. No adverse developmental effects were observed during organogenesis at up to 48-times (rats) and 128-times (rabbits) the maximum clinical dose; higher doses caused maternal and fetal toxicity. Reduced body weight was observed in offspring at exposures ≥4-times the maximum clinical dose.
Lactation
Risk Summary: Limited information is available on the presence of Empagliflozin in human milk, its effects on the breastfed infant, or on milk production. Empagliflozin is present in rat milk. There may be risk to the developing human kidney. Because of the potential for serious adverse reactions in a breastfed infant, use is not recommended while breastfeeding.
Data: Empagliflozin was present in rat milk and fetal tissues, with its accumulation in milk. Juvenile rats exposed to empagliflozin showed renal pelvic and tubular dilatations during maturation.
Paediatric Use
Safety and effectiveness of Empagliflozin as an adjunct to diet and exercise to improve glycaemic control in type 2 diabetes mellitus have been established in paediatric patients aged 10 years and older; safety profile was similar to adults, except hypoglycaemia risk was higher. Safety and effectiveness have not been established in patients less than 10 years of age or for reduction of cardiovascular or renal outcomes in paediatric patients.
Geriatric Use
Empagliflozin is expected to have diminished glycaemic efficacy in elderly patients with renal impairment. The risk of volume depletion-related adverse reactions and urinary tract infections increased in patients 75 years of age and older. No overall differences in safety and effectiveness were observed between patients 65 years of age and older and younger adult patients.
Renal Impairment
Glucose lowering benefit decreases with worsening renal function. Risks of renal impairment, volume depletion, and urinary tract infection-related adverse reactions increase. Use for glycemic control is not recommended when eGFR is less than 30 mL/min/1.73 m² in patients without established cardiovascular disease or risk factors. Safety profile across eGFR subgroups was consistent with the known safety profile.
Hepatic Impairment
Empagliflozin may be used in patients with hepatic impairment.
Empagliflozin has minor influence on the ability to drive and use machines. Patients should be advised to take precautions to avoid hypoglycaemia while driving and using machines, in particular when Empagliflozin is used in combination with a sulphonylurea and/or insulin.
The following important adverse reactions are described below:
Reporting of Suspected Adverse Reactions
Reporting suspected adverse reactions after authorization of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via email to: medico@zorvia.com
By reporting side effects, you can help provide more information on the safety of this medicine.
In controlled clinical studies single doses of up to 800 mg empagliflozin in healthy volunteers and multiple daily doses of up to 100 mg empagliflozin in patients with type 2 diabetes did not show any toxicity. Empagliflozin increased urine glucose excretion leading to an increase in urine volume. The observed increase in urine volume was not dose-dependent and is not clinically meaningful. There is no experience with doses above 800 mg in humans.
In the event of an overdose, treatment should be initiated as appropriate to the patient ‘s clinical status. The removal of empagliflozin by haemodialysis has not been studied.
Empagliflozin is an inhibitor of SGLT2, the predominant transporter responsible for reabsorption of glucose from the glomerular filtrate back into the circulation. By inhibiting SGLT2, empagliflozin reduces renal reabsorption of filtered glucose and lowers the renal threshold for glucose and thereby increases urinary glucose excretion.
Empagliflozin also reduces sodium reabsorption and increases the delivery of sodium to the distal tubule. This may influence several physiological functions including, but not restricted to, increasing tubuloglomerular feedback and reducing intraglomerular pressure, lowering both pre- and afterload of the heart and downregulating sympathetic activity.
Urinary Glucose Excretion
In patients with type 2 diabetes mellitus, urinary glucose excretion increased immediately following a dose of empagliflozin and was maintained at the end of a 4-week treatment period averaging at approximately 64 grams per day with 10 mg empagliflozin and 78 grams per day with 25 mg empagliflozin once daily. Data from single oral doses of empagliflozin in healthy subjects indicate that, on average, elevation in urinary glucose excretion approaches baseline by about 3 days for the 10 mg and 25 mg doses.
Urinary Volume
In a 5-day study, mean 24-hour urine volume increase from baseline was 341 mL on Day 1 and 135 mL on Day 5 of empagliflozin 25 mg once daily treatment.
Cardiac Electrophysiology
In a randomized, placebo-controlled, active-comparator, crossover study, 30 healthy subjects were administered a single oral dose of empagliflozin 25 mg, empagliflozin 200 mg (8 times the maximum dose), moxifloxacin, and placebo. No increase in QTc was observed with either 25 mg or 200 mg empagliflozin.
The pharmacokinetics of empagliflozin has been characterized in healthy volunteers and patients with type 2 diabetes mellitus and no clinically relevant differences were noted between the two populations. The steady state mean plasma AUC and Cmax were 1,870 nmol. h/L and 259 nmol/L, respectively, with 10 mg empagliflozin once daily treatment, and 4,740 nmol h/L and 687 nmol/L, respectively, with 25 mg empagliflozin once daily treatment. Systemic exposure of empagliflozin increased in a dose-proportional manner in the therapeutic dose range. Empagliflozin does not appear to have time-dependent pharmacokinetic characteristics. Following once daily dosing, up to 22% accumulation, with respect to plasma AUC, was observed at steady-state.
Absorption
After oral administration, peak plasma concentrations of empagliflozin were reached at 1.5 hours post-dose. Administration of 25 mg empagliflozin after intake of a high-fat and high-calorie meal resulted in slightly lower exposure; AUC decreased by approximately 16% and Cmax decreased by approximately 37%, compared to fasted condition. The observed effect of food on empagliflozin pharmacokinetics was not considered clinically relevant and empagliflozin may be administered with or without food.
Distribution
The apparent steady-state volume of distribution was estimated to be 73.8 L based on a population pharmacokinetic analysis. Following administration of an oral [14C]-empagliflozin solution to healthy subjects, the red blood cell partitioning was approximately 36.8% and plasma protein binding was 86.2%.
Elimination
The apparent terminal elimination half-life of empagliflozin was estimated to be 12.4 h and apparent oral clearance was 10.6 L/h based on the population pharmacokinetic analysis.
Metabolism
No major metabolites of empagliflozin were detected in human plasma and the most abundant metabolites were three glucuronide conjugates (2-O-, 3-O-, and 6-O-glucuronide). Systemic exposure of each metabolite was less than 10% of total drug-related material. In vitro studies suggested that the primary route of metabolism of empagliflozin in humans is glucuronidation by the uridine 5'-diphospho-glucuronosyltransferases UGT2B7, UGT1A3, UGT1A8, and UGT1A9.
Excretion
Following administration of an oral [14C]-empagliflozin solution to healthy subjects, approximately 95.6% of the drug-related radioactivity was eliminated in feces (41.2%) or urine (54.4%). The majority of drug-related radioactivity recovered in feces was unchanged parent drug and approximately half of drug-related radioactivity excreted in urine was unchanged parent drug.
Specific Populations
Pediatric Patients
The pharmacokinetics and pharmacodynamics of empagliflozin were investigated in pediatric patients aged 10 to 17 years with type 2 diabetes mellitus. Oral administration of empagliflozin at 10 mg and 25 mg resulted in exposure within the range observed in adult patients.
Effects of Age, Body Mass Index, Gender, and Race
Age, body mass index (BMI), gender and race (Asians versus primarily Whites) do not have a clinically meaningful effect on pharmacokinetics of empagliflozin.
Patients with Hepatic Impairment
In adult patients with mild, moderate, and severe hepatic impairment according to the Child-Pugh classification, AUC of empagliflozin increased by approximately 23%, 47%, and 75%, and Cmax increased by approximately 4%, 23%, and 48%, respectively, compared to subjects with normal hepatic function.
Patients with Renal Impairment
In adult patients with type 2 diabetes mellitus with mild (eGFR: 60 to less than 90 mL/min/1.73 m2), moderate (eGFR: 30 to less than 60 mL/min/1.73 m2), and severe (eGFR: less than 30 mL/min/1.73 m2) renal impairment and patients on dialysis due to kidney failure, AUC of empagliflozin increased by approximately 18%, 20%, 66%, and 48%, respectively, compared to subjects with normal renal function. Peak plasma levels of empagliflozin were similar in patients with moderate renal impairment and patients on dialysis due to kidney failure compared to subjects with normal renal function. Peak plasma levels of empagliflozin were roughly 20% higher in patients with mild and severe renal impairment as compared to patients with normal renal function. Population pharmacokinetic analysis showed that the apparent oral clearance of empagliflozin decreased, with a decrease in eGFR leading to an increase in drug exposure. However, the fraction of empagliflozin that was excreted unchanged in urine, and urinary glucose excretion, declined with decrease in eGFR.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenesis was evaluated in 2-year studies conducted in CD-1 mice and Wistar rats. Empagliflozin did not increase the incidence of tumors in female rats dosed at 100, 300, or 700 mg/kg/day (up to 72 times the exposure from the maximum clinical dose of 25 mg). In male rats, hemangiomas of the mesenteric lymph node were increased significantly at 700 mg/kg/day or approximately 42 times the exposure from a 25 mg clinical dose. Empagliflozin did not increase the incidence of tumors in female mice dosed at 100, 300, or 1,000 mg/kg/day (up to 62 times the exposure from a 25 mg clinical dose). Renal tubule adenomas and carcinomas were observed in male mice at 1,000 mg/kg/day, which is approximately 45 times the exposure of the maximum clinical dose of 25 mg. These tumors may be associated with a metabolic pathway predominantly present in the male mouse kidney.
Mutagenesis
Empagliflozin was not mutagenic or clastogenic with or without metabolic activation in the in vitro Ames bacterial mutagenicity assay, the in vitro L5178Y tk+/- mouse lymphoma cell assay, and an in vivo micronucleus assay in rats.
Impairment of Fertility
Empagliflozin had no effects on mating, fertility or early embryonic development in treated male or female rats up to the high dose of 700 mg/kg/day (approximately 155 times the 25 mg clinical dose in males and females, respectively).
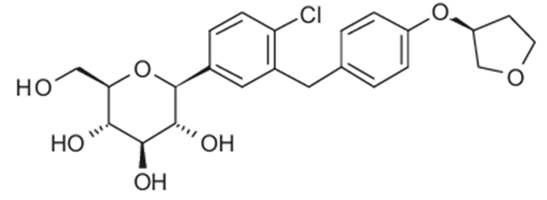
Emplic tablets for oral use contain empagliflozin, an inhibitor of the SGLT2.
Chemical name: D-Glucitol,1,5-anhydro-1-C-[4-chloro-3-[[4-[[(3S)-tetrahydro-3
furanyl]oxy]phenyl]methyl]phenyl]-, (1S).
Molecular formula: C23H27ClO7
Molecular weight: 450.91
Structural formula is:
None.
Refer on pack.
10×10 Tablet
Keep out of reach of children.
Store below 30°C in a dry place. Protect from light.
17th March 2026