4.4 Special Warnings and Precautions for use
General
Linectra-M 500/1000 should not be used in patients with type 1 diabetes.
Hypoglycaemia
When linagliptin was added to a sulphonylurea on a background of metformin, the incidence of hypoglycaemia was increased over that of placebo.
Sulphonylureas and insulin are known to cause hypoglycaemia. Therefore, caution is advised when Linectra-M 500/1000 is used in combination with a sulphonylurea and/or insulin. A dose reduction of the sulphonylurea or insulin may be considered.
Hypoglycaemia is not identified as adverse reaction for linagliptin, metformin, or linagliptin plus metformin. In clinical trials, the incidence rates of hypoglycemia were comparably low in patients taking linagliptin in combination with metformin or metformin alone.
Lactic acidosis
Lactic acidosis, a very rare but serious metabolic complication, most often occurs at acute worsening of renal function or cardiorespiratory illness or sepsis. Metformin accumulation occurs at acute worsening of renal function and increases the risk of lactic acidosis.
In case of dehydration (severe diarrhoea or vomiting, fever or reduced fluid intake), metformin should be temporarily discontinued and contact with a health care professional is recommended.
Medicinal products that can acutely impair renal function (such as antihypertensives, diuretics and NSAIDs) should be initiated with caution in metformin-treated patients. Other risk factors for lactic acidosis are excessive alcohol intake, hepatic impairment, inadequately controlled diabetes, ketosis, prolonged fasting and any conditions associated with hypoxia, as well as concomitant use of medicinal products that may cause lactic acidosis.
Patients and/or caregivers should be informed of the risk of lactic acidosis. Lactic acidosis is characterised by acidotic dyspnoea, abdominal pain, muscle cramps, asthenia and hypothermia followed by coma. In case of suspected symptoms, the patient should stop taking metformin and seek immediate medical attention. Diagnostic laboratory findings are decreased blood pH (< 7.35), increased plasma lactate levels (> 5 mmol/L) and an increased anion gap and lactate/pyruvate ratio.
Patients with known or suspected mitochondrial diseases
In patients with known mitochondrial diseases such as Mitochondrial Encephalopathy with Lactic Acidosis, and Stroke-like episodes (MELAS) syndrome and Maternal inherited diabetes and deafness (MIDD), metformin is not recommended due to the risk of lactic acidosis exacerbation and neurologic complications which may lead to worsening of the disease.
In case of signs and symptoms suggestive of MELAS syndrome or MIDD after the intake of metformin, treatment with metformin should be withdrawn immediately and prompt diagnostic evaluation should be performed.
Administration of iodinated contrast agent
Intravascular administration of iodinated contrast agents may lead to contrast induced nephropathy, resulting in metformin accumulation and an increased risk of lactic acidosis. Metformin should be discontinued prior to or at the time of the imaging procedure and not restarted until at least 48 hours after, provided that renal function has been re-evaluated and found to be stable.
Renal function
GFR should be assessed before treatment initiation and regularly thereafter. Metformin is contraindicated in patients with GFR< 30 mL/min and should be temporarily discontinued in the presence of conditions that alter renal function.
Cardiac function
Patients with heart failure are more at risk of hypoxia and renal impairment. In patients with stable chronic heart failure, Linectra-M 500/1000 may be used with a regular monitoring of cardiac and renal function.
For patients with acute and unstable heart failure, Linectra-M 500/1000 is contraindicated.
Surgery
Metformin must be discontinued at the time of surgery under general, spinal or epidural anesthesia. Therapy may be restarted no earlier than 48 hours following surgery or resumption of oral nutrition and provided that renal function has been re-evaluated and found to be stable.
Elderly
Caution should be exercised when treating patients 80 years and older
Change in clinical status of patients with previously controlled type 2 diabetes
As Linectra-M 500/1000 contains metformin, a patient with previously well controlled type 2 diabetes on Linectra-M 500/1000 who develops laboratory abnormalities or clinical illness (especially vague and poorly defined illness) should be evaluated promptly for evidence of ketoacidosis or lactic acidosis. Evaluation should include serum electrolytes and ketones, blood glucose and, if indicated, blood pH, lactate, pyruvate, and metformin levels. If acidosis of either form occurs, Linectra-M 500/1000 must be stopped immediately and other appropriate corrective measures initiated.
Acute pancreatitis
Use of DPP-4 inhibitors has been associated with a risk of developing acute pancreatitis. Acute pancreatitis has been observed in patients taking linagliptin. In a cardiovascular and renal safety study (CARMELINA) with median observation period of 2.2 years, adjudicated acute pancreatitis was reported in 0.3% of patients treated with linagliptin and in 0.1% of patients treated with placebo. Patients should be informed of the characteristic symptoms of acute pancreatitis. If pancreatitis is suspected, Linectra-M 500/1000 should be discontinued; if acute pancreatitis is confirmed, Linectra-M 500/1000 should not be restarted. Caution should be exercised in patients with a history of pancreatitis.
Bullous pemphigoid
Bullous pemphigoid has been observed in patients taking linagliptin. In the CARMELINA study, bullous pemphigoid was reported in 0.2% of patients on treatment with linagliptin and in no patient on placebo. If bullous pemphigoid is suspected, Linectra-M 500/1000 should be discontinued.
Vitamin B12
Metformin may reduce vitamin B12 levels. The risk of low vitamin B12 levels increases with increasing metformin dose, treatment duration, and/or in patients with risk factors known to cause vitamin B12 deficiency. In case of suspicion of vitamin B12 deficiency (such as anaemia or neuropathy), vitamin B12 serum levels should be monitored. Periodic vitamin B12 monitoring could be necessary in patients with risk factors for vitamin B12 deficiency. Metformin therapy should be continued for as long as it is tolerated and not contra-indicated and appropriate corrective treatment for vitamin B12 deficiency provided in line with current clinical guidelines.
4.5 Drug Interactions
Table 1: Clinically Relevant Interactions with Linectra-M 500/1000
|
Carbonic Anhydrase Inhibitors
|
|
Clinical Impact
|
Topiramate or other carbonic anhydrase inhibitors (e.g., zonisamide, acetazolamide or dichlorphenamide) frequently cause a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with Linectra-M 500/1000 may increase the risk of lactic acidosis.
|
|
Intervention
|
Consider more frequent monitoring of these patients.
|
|
Drugs that Reduce Metformin Clearance
|
|
Clinical Impact
|
Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g., organic cationic transporter-2 [OCT2]/multidrug and toxin extrusion [MATE] inhibitors such as ranolazine, vandetanib, dolutegravir, and cimetidine) could increase systemic exposure to metformin and may increase the risk for lactic acidosis
|
|
Intervention
|
Consider the benefits and risks of concomitant use.
|
|
Alcohol
|
|
|
Clinical Impact
|
Alcohol is known to potentiate the effect of metformin on lactate metabolism.
|
|
Intervention
|
Warn patients against excessive alcohol intake while receiving Linectra-M 500/1000
|
|
Insulin or Insulin Secretagogues
|
|
Clinical Impact
|
The risk of hypoglycemia is increased when Linectra-M 500/1000 is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin.
|
|
Intervention
|
Coadministration of Linectra-M 500/1000 with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower doses of the insulin secretagogue or insulin to reduce the risk of hypoglycemia.
|
|
Drugs Affecting Glycaemic Control
|
|
Clinical Impact
|
Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. These drugs include the thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blocking drugs, and isoniazid.
|
|
Intervention
|
When such drugs are administered to a patient receiving Linectra-M 500/1000, the patient should be closely observed to maintain adequate glycaemic control. When such drugs are withdrawn from a patient receiving Linectra-M 500/1000, the patient should be observed closely for hypoglycaemia.
|
|
Inducers of P-glycoprotein or CYP3A4 Enzymes
|
|
Clinical Impact
|
Rifampin decreased linagliptin exposure, suggesting that the efficacy of linagliptin may be reduced when administered in combination with a strong P-gp or CYP3A4 inducer.
|
|
Intervention
|
Use of alternative treatments is strongly recommended when linagliptin is to be administered with a strong P-gp or CYP3A4 inducer.
|
5.0 Pharmacological Properties
5.1 Mechanism of Action
Linectra-M 500/1000 contains: linagliptin, a dipeptidyl peptidase-4 (DPP-4) inhibitor, and metformin, a biguanide.
Linagliptin
Linagliptin is an inhibitor of DPP-4, an enzyme that degrades the incretin hormones glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). Thus, linagliptin increases the concentrations of active incretin hormones, stimulating the release of insulin in a glucose-dependent manner and decreasing the levels of glucagon in the circulation. Both incretin hormones are involved in the physiological regulation of glucose homeostasis. Incretin hormones are secreted at a low basal level throughout the day and levels rise immediately after meal intake. GLP-1 and GIP increase insulin biosynthesis and secretion from pancreatic beta cells in the presence of normal and elevated blood glucose levels. Furthermore, GLP-1 also reduces glucagon secretion from pancreatic alpha cells, resulting in a reduction in hepatic glucose output.
Metformin HCl
Metformin is an antihyperglycemic agent which improves glucose tolerance in patients with type 2 diabetes mellitus, lowering both basal and postprandial plasma glucose. Metformin decreases hepatic glucose production, decreases intestinal absorption of glucose, and improves insulin sensitivity by increasing peripheral glucose uptake and utilization. With metformin therapy, insulin secretion remains unchanged while fasting insulin levels and day-long plasma insulin response may decrease.
5.2 Pharmacodynamic Properties
Linagliptin
Linagliptin binds to DPP-4 in a reversible manner and increases the concentrations of incretin hormones. Linagliptin glucose-dependently increases insulin secretion and lowers glucagon secretion, thus resulting in a better regulation of the glucose homeostasis. Linagliptin binds selectively to DPP-4 and selectively inhibits DPP-4, but not DPP-8 or DPP-9 activity in vitro at concentrations approximating therapeutic exposures.
Cardiac Electrophysiology
In a randomized, placebo-controlled, active-comparator, 4-way crossover study, 36 healthy subjects were administered a single oral dose of linagliptin 5 mg, linagliptin 100 mg (20 times the recommended dose), moxifloxacin, and placebo. No increase in QTc was observed with either the recommended dose of 5 mg or the 100-mg dose. At the 100-mg dose, peak linagliptin plasma concentrations were approximately 38-fold higher than the peak concentrations following a 5-mg dose.
5.3 Pharmacokinetic Properties
Absorption
Linagliptin
The absolute bioavailability of linagliptin is approximately 30%. Following oral administration, plasma concentrations of linagliptin decline in at least a biphasic manner with a long terminal half-life (>100 hours), related to the saturable binding of linagliptin to DPP-4. However, the prolonged elimination does not contribute to the accumulation of the drug. The effective half-life for accumulation of linagliptin, as determined from oral administration of multiple doses of linagliptin 5 mg, is approximately 12 hours. After once-daily dosing, steady-state plasma concentrations of linagliptin 5 mg are reached by the third dose, and Cmax and AUC increased by a factor of 1.3 at steady-state compared with the first dose. Plasma AUC of linagliptin increased in a less than dose-proportional manner in the dose range of 1 to 10 mg. The pharmacokinetics of linagliptin is similar in healthy subjects and in patients with type 2 diabetes mellitus.
Metformin HCl
Following a single oral dose of 1,000 mg (2 x 500 mg tablets) metformin extended release after a meal, the time to reach maximum plasma metformin concentration (Tmax) is achieved at approximately 7 to 8 hours. In both single- and multiple-dose studies in healthy subjects, once daily 1,000 mg (2 x 500 mg tablets) dosing provides equivalent systemic exposure, as measured by AUC, and up to 35% higher Cmax of metformin relative to the immediate-release given as 500 mg twice daily.
Single oral doses of metformin extended release from 500 mg to 2,500 mg resulted in less than proportional increase in both AUC and Cmax. Low-fat and high-fat meals increased the systemic exposure (as measured by AUC) from metformin extended-release tablets by about 38% and 73%, respectively, relative to fasting. Both meals prolonged metformin Tmax by approximately 3 hours but Cmax was not affected.
Distribution
Linagliptin
The mean apparent volume of distribution at steady-state following a single intravenous dose of linagliptin 5 mg to healthy subjects is approximately 1,110 L, indicating that linagliptin extensively distributes to the tissues. Plasma protein binding of linagliptin is concentration-dependent decreasing from about 99% at 1 nmol/L to 75% to 89% at ≥30 nmol/L, reflecting saturation of binding to DPP-4 with increasing concentration of linagliptin. At high concentrations, where DPP-4 is fully saturated, 70% to 80% of linagliptin remains bound to plasma proteins and 20% to 30% is unbound in plasma. Plasma binding is not altered in patients with renal or hepatic impairment.
Metformin HCl
The apparent volume of distribution (V/F) of metformin following single oral doses of immediate-release metformin HCl tablets 850 mg averaged 654±358 L. Metformin is negligibly bound to plasma proteins. Metformin partitions into erythrocytes, most likely as a function of time.
Elimination
Linagliptin: Linagliptin has a terminal half-life of about 200 hours at steady-state, though the accumulation half-life is about 11 hours. Renal clearance at steady-state was approximately 70 mL/min.
Metformin HCl: Metformin has a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
Metabolism
Linagliptin: Following oral administration, the majority (about 90%) of linagliptin is excreted unchanged, indicating that metabolism represents a minor elimination pathway. A small fraction of absorbed linagliptin is metabolized to a pharmacologically inactive metabolite, which shows a steady-state exposure of 13.3% relative to linagliptin.
Metformin HCl: Intravenous single-dose studies in normal subjects demonstrate that metformin does not undergo hepatic metabolism (no metabolites have been identified in humans), nor biliary excretion.
Excretion
Linagliptin: Following administration of an oral [14C] linagliptin dose to healthy subjects, approximately 85% of the administered radioactivity was eliminated via the enterohepatic system (80%) or urine (5%) within 4 days of dosing.
Metformin HCl: Following oral administration, approximately 90% of the absorbed drug is excreted via the renal route within the first 24 hours. Renal clearance is approximately 3.5 times greater than creatinine clearance, which indicates that tubular secretion is the major route of metformin elimination.
6.0 Nonclinical Properties
6.1 Animal Toxicology or Pharmacology
Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity, mutagenicity, or impairment of fertility studies have been conducted with the combination of linagliptin and metformin HCl.
Linagliptin
Linagliptin did not increase the incidence of tumors in male and female rats in a 2-year study at doses of 6, 18, and 60 mg/kg. The highest dose of 60 mg/kg is approximately 418 times the clinical dose of 5 mg/day based on AUC exposure. Linagliptin did not increase the incidence of tumors in mice in a 2-year study at doses up to 80 mg/kg (males) and 25 mg/kg (females), or approximately 35 and 270 times the clinical dose based on AUC exposure. Higher doses of linagliptin in female mice (80 mg/kg) increased the incidence of lymphoma at approximately 215 times the clinical dose based on AUC exposure.
Linagliptin was not mutagenic or clastogenic with or without metabolic activation in the Ames bacterial mutagenicity assay, a chromosomal aberration test in human lymphocytes, and an in vivo micronucleus assay.
In fertility studies in rats, linagliptin had no adverse effects on early embryonic development, mating, fertility, or bearing live young up to the highest dose of 240 mg/kg (approximately 943 times the clinical dose based on AUC exposure).
Metformin HCl
Long-term carcinogenicity studies have been performed in Sprague Dawley rats at doses of 150, 300, and 450 mg/kg/day in males and 150, 450, 900, and 1,200 mg/kg/day in females. These doses are approximately 2, 4, and 8 times in males, and 3, 7, 12, and 16 times in females of the maximum recommended human daily dose of 2,000 mg/kg/day based on body surface area comparisons. No evidence of carcinogenicity with metformin was found in either male or female rats. A carcinogenicity study was also performed in Tg.AC transgenic mice at doses of up to 2,000 mg/kg/day applied dermally. No evidence of carcinogenicity was observed in male or female mice.
Genotoxicity assessments in the Ames test, gene mutation test (mouse lymphoma cells), chromosomal aberrations test (human lymphocytes) and in vivo mouse micronucleus tests were negative.
Fertility of male or female rats was unaffected by metformin when administered at doses as high as 600 mg/kg/day, which is approximately 2 times the MRHD based on body surface area comparisons.
7.0 Description
Linectra-M 500/1000 tablets for oral use contain linagliptin and metformin HCl.
Linagliptin
Linagliptin is an inhibitor of the dipeptidyl peptidase-4 (DPP-4) enzyme
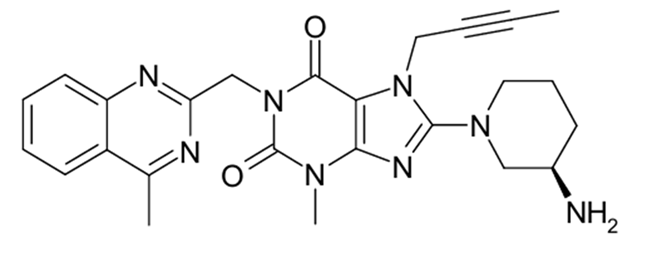
Chemical name: 1H-Purine-2,6-dione, 8-[(3R)-3-amino-1-piperidinyl]-7-(2-butyn-1-yl)-3,7-dihydro-3-methyl-1-[(4-methyl-2-quinazolinyl)methyl]-
Molecular formula: C25H28N8O2
Molecular weight: 472.54 g/mol.
Structural formula is:
Metformin HCl
Metformin HCl is a biguanide.
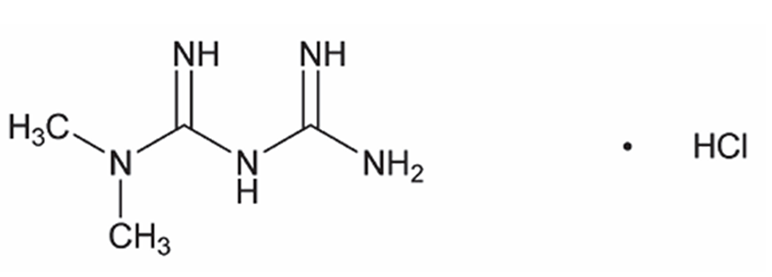
Chemical name: N,N-dimethylimidodicarbonimidic diamide HCl
Molecular formula: C4H11N5•HCl
Molecular weight: 165.63.
The structural formula is:
8.0 Pharmaceutical Particulars
8.1 Incompatibilities
None.
8.2 Shelf-Life
Refer on pack.
8.3 Packaging Information
As per carton.
8.4 Storage and Handling Instructions
Keep out of reach of children. Store below 30°C in a dry place. Protect from light.
9.0 Patient Counselling Information
Lactic Acidosis: Inform patients of the risks of lactic acidosis due to metformin, its symptoms, and conditions that predispose to its development. Advise patients to discontinue Linectra-M 500/1000 immediately and to notify their healthcare provider promptly if unexplained hyperventilation, malaise, myalgia, unusual somnolence, or other nonspecific symptoms occur. Counsel patients against excessive alcohol intake and inform patients about importance of regular testing of renal function while receiving Linectra-M 500/1000. Instruct patients to inform their healthcare provider that they are taking Linectra-M 500/1000 prior to any surgical or radiological procedure.
Pancreatitis: Inform patients that acute pancreatitis has been reported during use of linagliptin. Persistent severe abdominal pain, sometimes radiating to the back, which may or may not be accompanied by vomiting, is the hallmark symptom of acute pancreatitis. Instruct patients to discontinue Linectra-M 500/1000 promptly and contact their healthcare provider if persistent severe abdominal pain occurs.
Hypoglycaemia with Concomitant Use with Insulin and Insulin Secretagogues: Inform patients that the risk of hypoglycaemia is increased when Linectra-M 500/1000 is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin.
Hypersensitivity Reactions: Inform patients that serious allergic reactions, such as anaphylaxis, angioedema, and exfoliative skin conditions, have been reported with use of linagliptin. If symptoms of allergic reactions occur, patients must stop taking Linectra-M 500/1000 and seek medical advice promptly.
Administration Instructions: Inform patients that the tablets must be swallowed whole and never split, crushed, dissolved, or chewed and that incompletely dissolved tablets may be eliminated in the faeces.
Vitamin B12 Deficiency: Inform patients about the importance of regular haematological parameters while receiving Linectra-M 500/1000.
Severe and Disabling Arthralgia: Inform patients that severe and disabling joint pain may occur with this class of drugs. Instruct patients to seek medical advice if severe joint pain occurs.
Bullous Pemphigoid: Inform patients that bullous pemphigoid has been reported during use of linagliptin. Instruct patients to seek medical advice if blisters or erosions occur.
Heart Failure: Inform patients of the signs and symptoms of heart failure. Instruct patients to contact their healthcare provider as soon as possible if they experience symptoms of heart failure, including increasing shortness of breath, rapid increase in weight or swelling of the feet.
Patients of Reproductive Potential: Inform patients that treatment with metformin may result in ovulation in some premenopausal anovulatory patients, which may lead to unintended pregnancy.
Missed Dose: Instruct patients to take Linectra-M 500/1000 only as prescribed. If a dose is missed, it should be taken as soon as the patient remembers. Advise patients not to double their next dose.




